0. Quick Overview
ISO 10121 is the international standard series for gas-phase air cleaning media and devices (GPACM / GPACD) used in general ventilation systems. While particulate air filters remove solid and liquid aerosol particles from the air, molecular filters target gaseous contaminants – volatile organic compounds (VOCs), odours, corrosive gases, ozone, nitrogen dioxide, sulphur dioxide and a wide range of other airborne molecular pollutants that pass straight through even the highest-grade HEPA filter.
The standard series was developed under ISO Technical Committee 142 (Cleaning Equipment for Air and Other Gases) and consists of three parts. ISO 10121-3:2022 is the most recent and arguably the most significant addition: it introduces the first internationally standardised classification system for molecular filters treating outdoor air – comparable to what ISO 16890 achieved for particulate filters and what ISO 29463 established for HEPA and ULPA filters.
Current editions of the standard:
| Standard / Part | Content | Current Edition |
|---|---|---|
| ISO 10121-1 | Test method for gas-phase air cleaning media (GPACM) | 2014 (revision in progress: ISO/DIS 10121-1) |
| ISO 10121-2 | Test method for gas-phase air cleaning devices (GPACD) | 2013 (revision in progress: ISO/CD 10121-2) |
| ISO 10121-3 | Classification system for GPACDs applied to treatment of outdoor air | 2022 (Oct. 2022) |
1. Why Molecular Filtration Matters: Beyond Particulate Removal
Modern ventilation systems are designed to supply clean outdoor air to occupied spaces – offices, hospitals, schools, data centres, museums, industrial facilities. Particulate air filters to ISO 16890 or EN 1822 / ISO 29463 remove solid particles effectively, but they are essentially transparent to gaseous pollutants. In urban environments, the outdoor air delivered by a ventilation system routinely contains significant concentrations of nitrogen dioxide (NO2), ozone (O3), sulphur dioxide (SO2), volatile organic compounds (VOCs including toluene, benzene, formaldehyde) and a wide spectrum of odorous molecules.
These gaseous contaminants have measurable impacts on human health, productivity and comfort. NO2 and O3 are respiratory irritants linked to asthma exacerbation and reduced lung function. VOCs contribute to sick building syndrome and are associated with headaches, fatigue and impaired cognitive performance. In sensitive environments – semiconductor fabrication, museum conservation, data centres with electronic equipment – even trace concentrations of corrosive gases can cause irreversible damage to materials, artworks and electronic components.
Molecular filtration addresses this gap. Using activated carbon, impregnated carbon, chemisorptive substrates, catalytic media or micro ion exchange resins, molecular filters capture and neutralise gaseous pollutants through physical adsorption, chemical reaction or ion exchange. ISO 10121 provides the test and classification framework that makes the performance of these diverse technologies comparable, measurable and specifiable.

Molecular air filtration protects workers from harmful gas-phase contaminants including welding fumes, VOCs and industrial odours. ISO 10121 provides the standardised test framework for these filter systems.
2. Structure of ISO 10121 – All Three Parts
Part 1: Gas-Phase Air Cleaning Media – GPACM (ISO 10121-1:2014)
Part 1 defines the laboratory test method for evaluating the performance of gas-phase air cleaning media (GPACM) – the raw filtration material before it is assembled into a complete device. The standard identifies three types of media:
| Media Type | Abbreviation | Description | Typical Applications |
|---|---|---|---|
| Loose Fill | GPACM-LF | Granular or pelletised media (e.g. activated carbon granules, impregnated pellets) of various shapes and sizes, filled into trays, cartridges or deep-bed modules | Deep-bed adsorbers, refillable cartridge systems, packed-bed reactors |
| Flat Sheet | GPACM-FL | Flat fabric or nonwoven sheets containing or coated with adsorbent material; used as single layers, pleated or in bag-type configurations | Combined particulate/molecular filters, pleated panels, bag filters with carbon layer |
| Three-dimensional Structure | GPACM-TS | Structured media elements significantly thicker than flat sheet (e.g. honeycomb structures, corrugated media blocks, bonded carbon panels) | V-bank molecular filters, compact cassettes, modular adsorber systems |
The test procedure challenges the media with a defined test gas at controlled concentration, temperature, humidity and face velocity. The resulting performance curve shows how the removal efficiency declines over time as the media's adsorption capacity is consumed. This breakthrough behaviour is the fundamental characteristic of any molecular filter medium – unlike particulate filters, which become more efficient as they load with dust, molecular filters become less efficient as their capacity is exhausted.
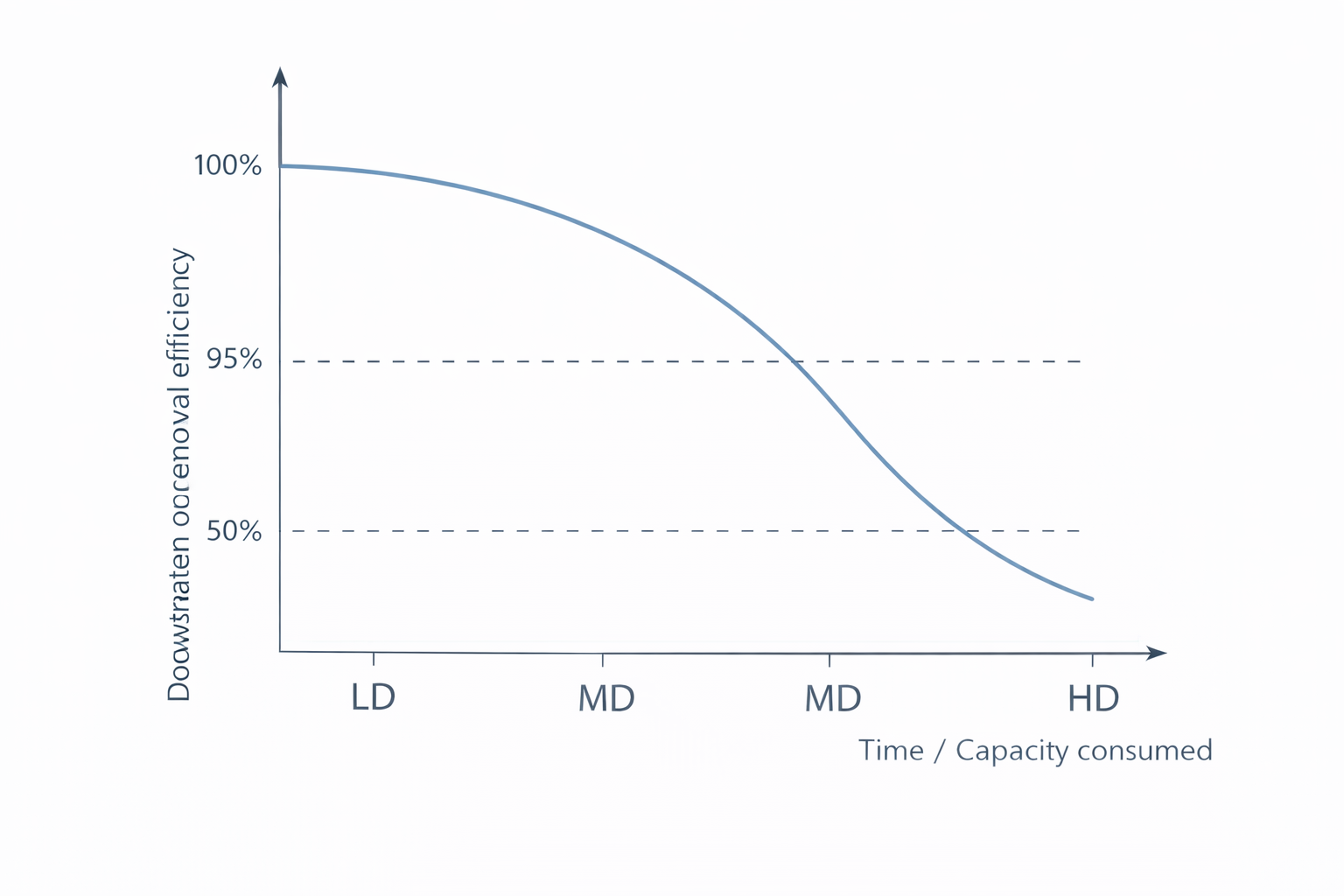
The breakthrough curve is the characteristic performance profile of any molecular filter: removal efficiency starts high but declines progressively as the adsorption capacity is consumed. The ISO 10121-3 duty levels (vLD, LD, MD, HD) correspond to defined capacity thresholds on this curve – the higher the duty level, the longer the filter maintains effective gas removal.
A revision of Part 1 is currently under development (ISO/DIS 10121-1) to align it with the classification system introduced in Part 3.
Part 2: Gas-Phase Air Cleaning Devices – GPACD (ISO 10121-2:2013)
Part 2 extends the test methodology from media to complete gas-phase air cleaning devices (GPACD) – the assembled filter unit as installed in the ventilation system. This includes all practical aspects that influence real-world performance: filter housing design, media packing density, air distribution, bypass leakage, seal integrity and pressure drop.
The standard is intentionally technology-agnostic: it can be applied to activated carbon filters, impregnated carbon devices, chemisorptive media systems, catalytic converters, scrubbers, absorbers and even non-sorptive devices, provided they fit into the test apparatus and are intended for general ventilation applications.
A revision of Part 2 is also in progress (ISO/CD 10121-2).
Part 3: Classification System for Outdoor Air Treatment (ISO 10121-3:2022)
Part 3 is the landmark addition to the series. Published in October 2022, ISO 10121-3 establishes the first international classification system for molecular filters supplying single-pass outdoor air to general ventilation systems. For the first time, engineers, facility managers and specifiers can compare molecular filter performance using a standardised, manufacturer-independent rating system – just as ISO 16890 enables comparison of particulate filters.
The classification is based on four reference pollutant gases, each chosen because of its relevance to outdoor air quality in urban environments:
| Reference Pollutant | Formula | Category | Relevance |
|---|---|---|---|
| Ozone | O3 | Oxidising gas | Photochemical smog, respiratory irritant, material degradation |
| Sulphur dioxide | SO2 | Acidic gas | Industrial / combustion emissions, corrosion, acid deposition |
| Nitrogen dioxide | NO2 | Acidic / oxidising gas | Traffic emissions, respiratory hazard, material corrosion |
| Toluene | C7H8 | VOC (representative) | Represents the broad group of volatile organic compounds |
For each pollutant, the filter is tested and classified according to two dimensions: average efficiency and capacity (duty level).
3. The ISO 10121-3 Classification Code Explained
The classification result is expressed in a standardised format that combines pollutant name, duty level and average efficiency. An example classification reads:
Toluene MD 90
This means: tested against toluene, the filter achieves Medium Duty capacity with a 90 % average removal efficiency over its rated service life.
Duty Levels (Capacity Classes)
ISO 10121-3 defines four duty levels that reflect the filter's service life – the amount of pollutant (in mol/m²) the filter can absorb before its efficiency drops below 50 %:
| Duty Level | Abbreviation | Capacity Threshold | Relative Service Life |
|---|---|---|---|
| Very Light Duty | vLD | 0.75 mol/m² | Entry-level (50 % of LD) |
| Light Duty | LD | 1.5 mol/m² | Baseline |
| Medium Duty | MD | 6.0 mol/m² | 4 × LD |
| Heavy Duty | HD | 24.0 mol/m² | 16 × LD |
The factor of four between each step is deliberate: moving from LD to MD quadruples the filter's capacity, and from MD to HD quadruples it again. A Heavy Duty filter therefore has 16 times the capacity of a Light Duty filter – corresponding to dramatically longer service intervals and lower life-cycle costs.
Average Efficiency
The average removal efficiency is measured over the entire rated service life of the filter and rounded down in 5 % increments. A filter achieving 92 % average efficiency is thus classified as "90". The minimum reportable efficiency is 50 %; filters dropping below this threshold during the initial low-concentration screening phase are disqualified from further testing.
Complete Classification Example
A molecular filter might receive the following full ISO 10121-3 classification:
| Pollutant | Classification | Interpretation |
|---|---|---|
| O3 | O3 HD 85 | Heavy Duty capacity, 85 % avg. efficiency for ozone |
| SO2 | SO2 MD 75 | Medium Duty capacity, 75 % avg. efficiency for SO2 |
| NO2 | NO2 MD 70 | Medium Duty capacity, 70 % avg. efficiency for NO2 |
| Toluene | Toluene HD 90 | Heavy Duty capacity, 90 % avg. efficiency for toluene/VOC |
This multi-pollutant classification allows specifiers to select filters tailored to their specific air quality challenge – a building near a major road (NO2 dominant) requires a different filter profile than an industrial site (SO2, VOCs) or a high-altitude location with elevated ozone.
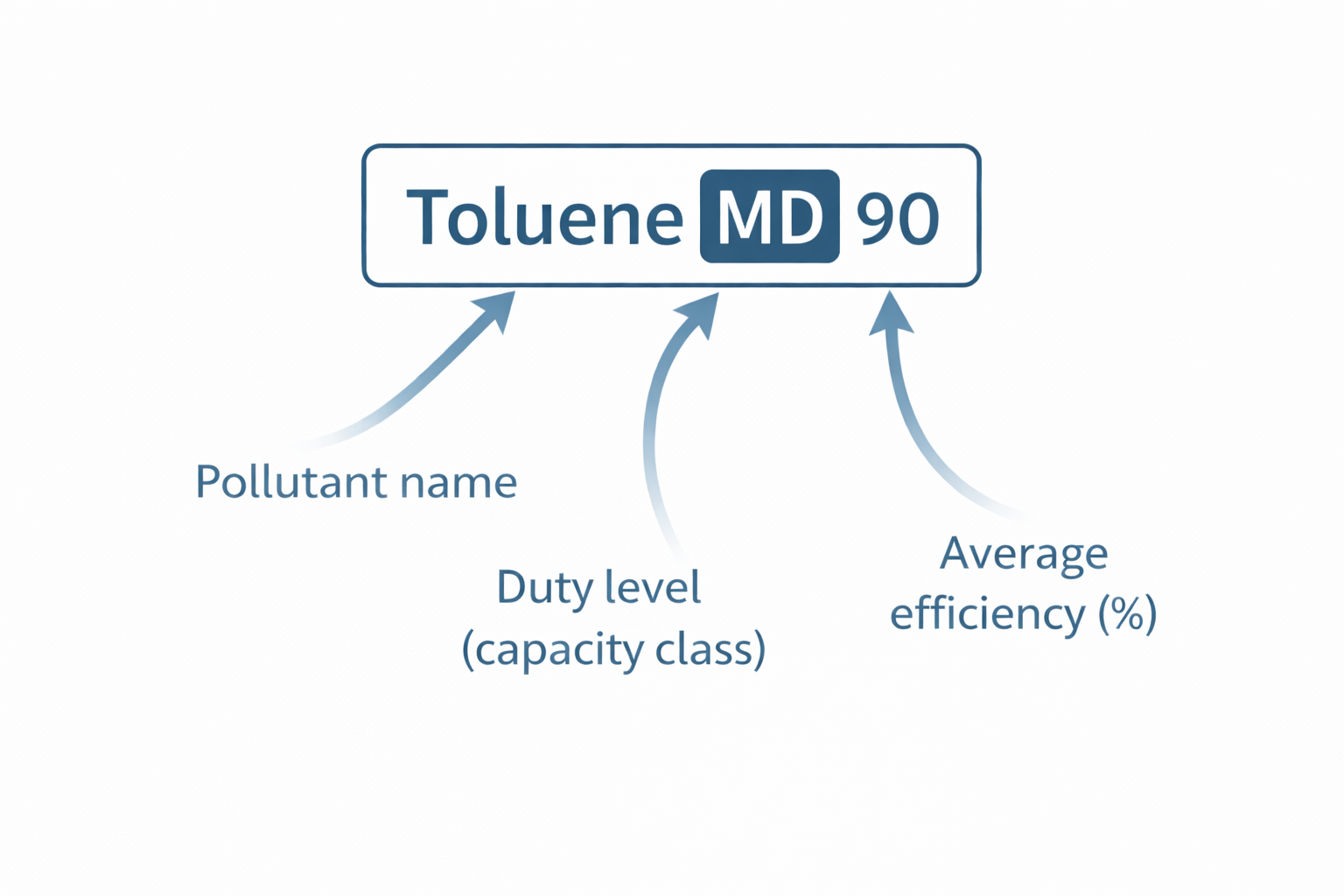
The ISO 10121-3 classification label combines three elements: the reference pollutant (e.g. Toluene), the duty level (vLD / LD / MD / HD) indicating the filter's capacity, and the average removal efficiency in percent. This standardised format enables direct, manufacturer-independent comparison of molecular filter performance.
4. The Science of Gas-Phase Air Cleaning: How Molecular Filters Work
Molecular air filters remove gaseous contaminants through fundamentally different mechanisms than particulate filters. While particulate filters intercept solid and liquid particles through physical barriers (sieving, impaction, diffusion, interception), molecular filters capture gas molecules through surface interactions at the molecular level. The three principal mechanisms are physical adsorption (physisorption), chemical adsorption (chemisorption) and ion exchange.
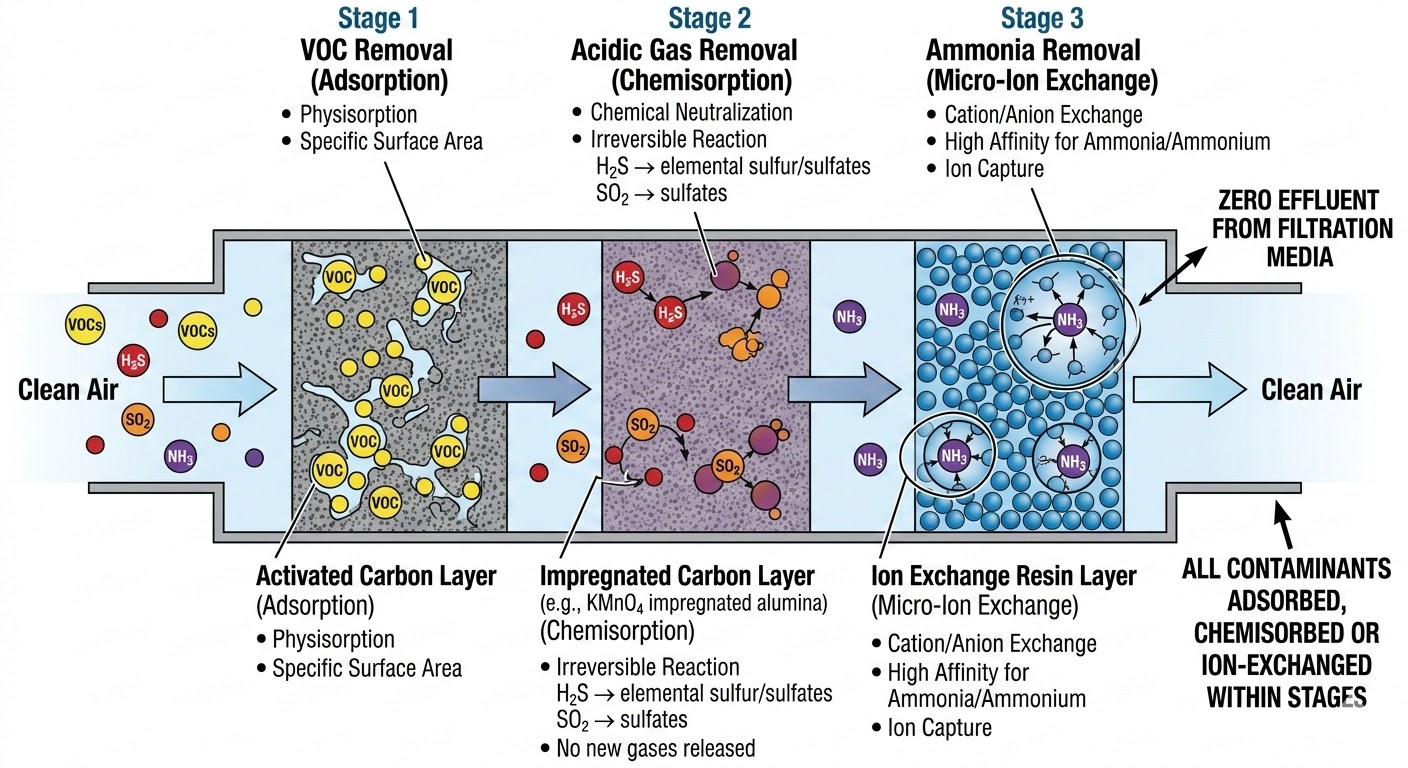
The three principal mechanisms of molecular gas-phase air cleaning: physisorption (van der Waals bonding on activated carbon), chemisorption (irreversible chemical reaction on impregnated carbon or reactive substrates) and micro ion exchange (selective ion swapping on polymer resins). Understanding these mechanisms is essential for selecting the right media for a given pollutant profile under ISO 10121.
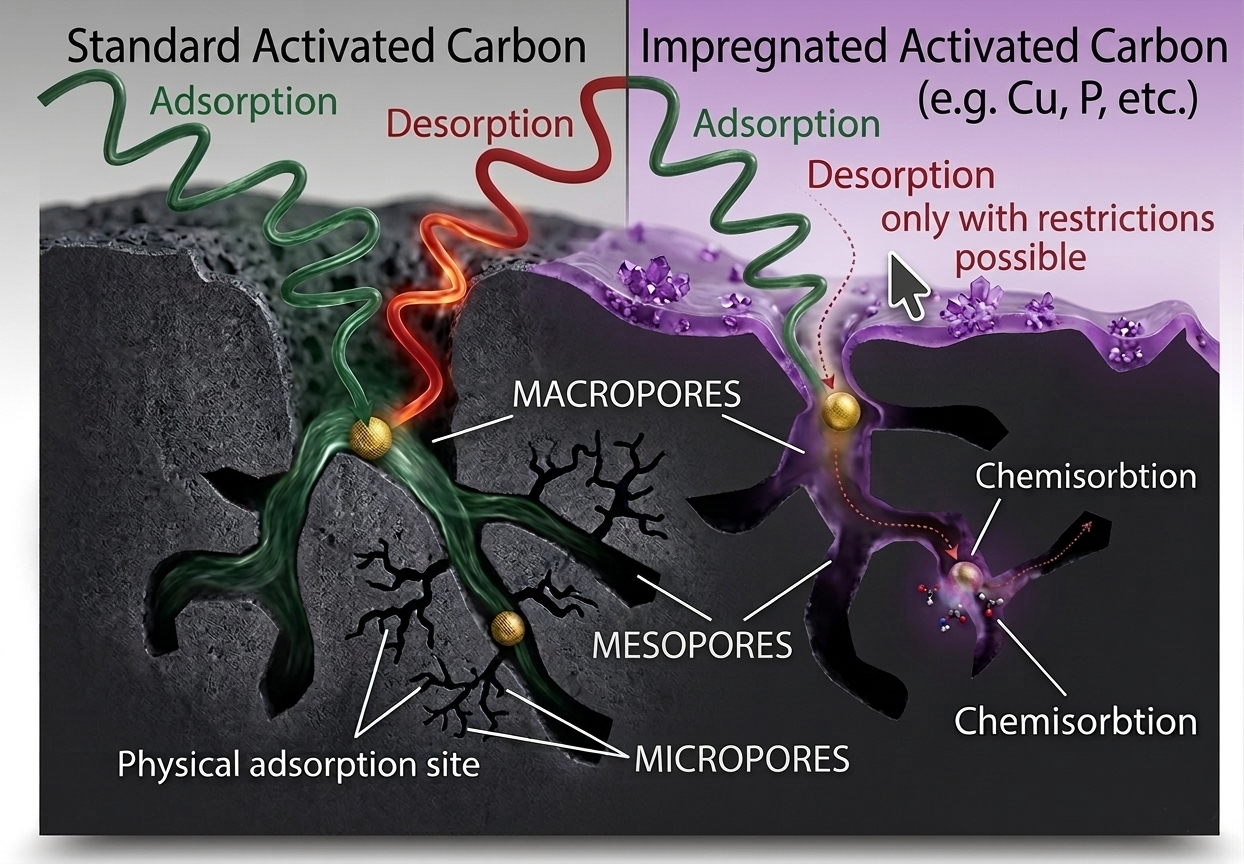
A closer look at the two carbon-based mechanisms: in physisorption (left), gas molecules are held by weak van der Waals forces and can potentially desorb; in chemisorption (right), the impregnant reacts with the gas molecule to form a stable, non-volatile compound that remains permanently bound in the pore.
4.1 Activated Carbon – The Universal Adsorbent

Activated carbon granules (here: 3 mm pelletised form) are the most widely used adsorbent in molecular air filtration. Their enormous internal surface area – typically 800 to 1,200 m² per gram – provides billions of microscopic binding sites for gas molecules.
Activated carbon is the most widely used adsorbent material in gas-phase air filtration. It is produced from carbonaceous raw materials – coconut shell, bituminous coal, wood or peat – through a two-stage process of carbonisation (thermal decomposition in the absence of oxygen) and activation (controlled oxidation with steam or CO2 at 800–1,000 °C). The activation process creates an extraordinarily porous internal structure with a specific surface area of 800 to 1,200 m²/g – a single gram of activated carbon can have an internal surface area equivalent to several football pitches.
This vast surface area is distributed across three types of pores: micropores (< 2 nm), mesopores (2–50 nm) and macropores (> 50 nm). The micropores account for the majority of the adsorptive capacity and are responsible for capturing small gas molecules through physical adsorption (physisorption) – a process driven by weak van der Waals forces between the carbon surface and the gas molecule. The molecule is not chemically altered; it is held on the surface by intermolecular attraction and can, in principle, be released (desorbed) if conditions change.
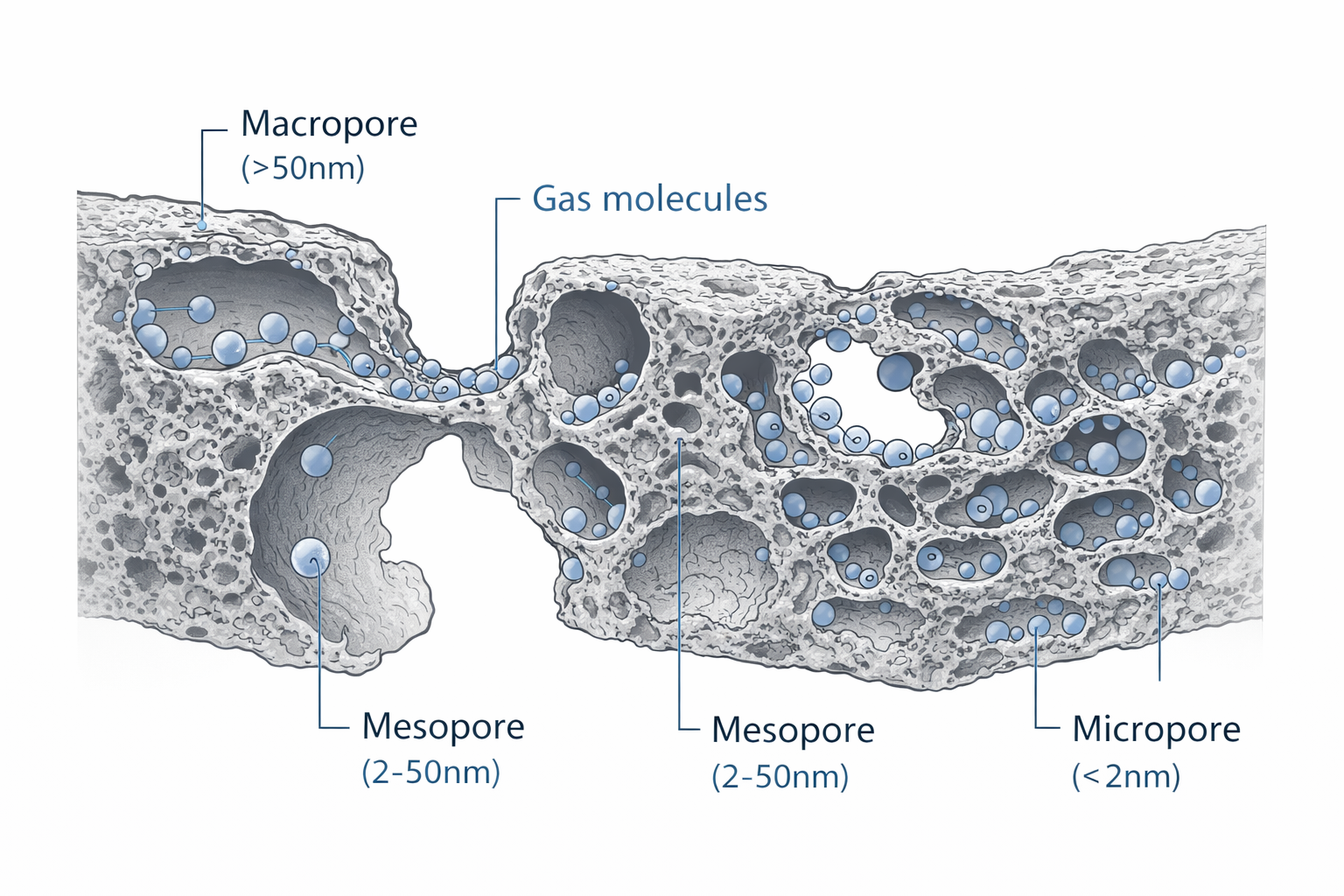
The hierarchical pore structure of activated carbon: macropores (> 50 nm) act as transport channels, mesopores (2–50 nm) provide intermediate access paths, and micropores (< 2 nm) deliver the vast majority of the adsorptive surface area where gas molecules are captured. The pore size distribution determines which molecules can be adsorbed and at what capacity.
Activated carbon is particularly effective against non-polar and weakly polar organic molecules: volatile organic compounds (VOCs) such as toluene, xylene, benzene, styrene, formaldehyde; odorous compounds including mercaptans, amines, aldehydes; and many industrial solvents. Its broad-spectrum capability makes it the backbone of most molecular filtration systems. However, standard (unimpregnated) activated carbon has limited effectiveness against small, polar, inorganic molecules such as SO2, NO2, H2S, NH3 and formaldehyde at low concentrations – these require enhanced media.
4.2 Impregnated Activated Carbon – Targeted Chemical Enhancement
To extend the capability of activated carbon beyond physisorption, the carbon surface can be treated with chemical impregnants – reactive substances that are deposited within the pore structure during manufacturing. When a target gas molecule enters an impregnated pore, it reacts chemically with the impregnant and is converted into a harmless, non-volatile reaction product that remains permanently bound within the pore. This is chemisorption – an irreversible chemical binding process that is fundamentally different from physical adsorption.
Common impregnation chemistries include:
| Impregnant | Target Gases | Mechanism |
|---|---|---|
| Potassium hydroxide (KOH) | H2S, SO2, HCl, HF, Cl2 | Acid-base neutralisation of acidic gases |
| Phosphoric acid (H3PO4) | NH3, amines, basic gases | Acid-base neutralisation of alkaline gases |
| Potassium permanganate (KMnO4) | H2S, SO2, formaldehyde, NOx | Oxidation of reducing gases |
| Potassium iodide (KI) | Mercury vapour (Hg), radioactive iodine | Chemical complexation |
| Metal oxides (CuO, ZnO, MnO2) | H2S, mercaptans, arsine | Chemical reaction and catalytic oxidation |
Impregnation increases the effective capacity for target gases by a factor of 5 to 10 compared to unimpregnated carbon, but it comes at a trade-off: the impregnant occupies pore volume, reducing the carbon's capacity for broad-spectrum VOC adsorption. Effective molecular filter design therefore often combines impregnated and unimpregnated carbon layers or blends in a single device, optimised for the specific pollutant profile of the application.
4.3 Chemisorptive Substrates – Beyond Activated Carbon
A further category of gas-phase air cleaning media goes beyond carbon-based adsorption entirely. Chemisorptive substrates are engineered materials – typically porous ceramic, alumina or polymer carriers – that are loaded with highly reactive chemical compounds designed to target specific gas families with exceptional selectivity and capacity.
A notable example is the HS-Clean Pro range from HS-Luftfilterbau GmbH, which uses proprietary chemisorptive granulate media. These substrates are specifically engineered for the removal of acidic gases (SO2, NO2, HCl, HF), alkaline gases (NH3, amines), formaldehyde and other low-molecular-weight aldehydes. The chemisorptive reaction is irreversible and highly selective, delivering consistent removal performance even at the very low concentrations (ppb range) typical of outdoor air pollution – a regime where standard activated carbon may show poor efficiency.
Chemisorptive substrates are particularly valuable in applications where specific target gases must be removed reliably and where the risk of desorption (release of previously adsorbed molecules due to temperature or humidity changes) must be minimised. Because the binding is chemical rather than physical, chemisorptive media do not release captured pollutants under normal operating conditions – a significant safety advantage over physisorptive carbon in critical environments.
4.4 Micro Ion Exchange Resins – Selective Molecular Capture
Micro ion exchange resins represent the most selective class of gas-phase air cleaning media. These are polymer-based beads or granules with chemically functionalised surfaces that capture target gas molecules through ion exchange – replacing a harmless ion on the resin surface with a pollutant ion from the air stream.
The mechanism is highly specific: cation exchange resins target alkaline gases (NH3, amines), while anion exchange resins target acidic gases (SO2, NO2, HCl). The exchange reaction is fast, selective and effective even at extremely low gas concentrations. Micro ion exchange media complement activated carbon by addressing polar, low-molecular-weight gases that carbon handles poorly.
In practice, advanced molecular filter systems combine multiple media types in a single device – for example, an activated carbon layer for broad-spectrum VOC removal, an impregnated carbon layer for specific inorganic gases, and a micro ion exchange layer for ammonia or SO2. This multi-media approach delivers comprehensive gas-phase air cleaning across the full spectrum of outdoor and indoor pollutants.
| Media Type | Mechanism | Best For | Limitations |
|---|---|---|---|
| Activated carbon (unimpregnated) | Physisorption (van der Waals) | VOCs, odours, organic solvents, ozone | Limited for small polar molecules (NH3, formaldehyde, SO2 at low ppb); reversible (desorption risk) |
| Impregnated carbon | Chemisorption + physisorption | H2S, SO2, HCl, NH3, Hg, formaldehyde (depending on impregnant) | Reduced VOC capacity due to impregnant; specific to target gas family |
| Chemisorptive substrates | Irreversible chemical reaction | Acidic gases, alkaline gases, formaldehyde, low-ppb applications | Not broad-spectrum; engineered for specific pollutant families |
| Micro ion exchange | Ion exchange (selective) | NH3, amines (cation); SO2, NO2, HCl (anion) | Very specific; not effective for non-ionic molecules (VOCs) |
5. Applications – Where Molecular Filters to ISO 10121 Are Used
| Application | Key Contaminants | Typical Media | Relevant Standard |
|---|---|---|---|
| Commercial buildings, offices, schools | NO2, O3, VOCs, traffic-related odours | Activated carbon (V-bank or panel), combined particulate/molecular | ISO 10121-3, Eurovent 4/26 |
| Hospitals and healthcare | Anaesthetic gases, formaldehyde, disinfectant vapours, NO2 | Impregnated carbon, multi-media devices | ISO 10121-3, HTM 03-01 |
| Museums, archives, galleries | SO2, NO2, O3, acetic acid, formaldehyde | Impregnated carbon, chemisorptive substrates, ion exchange | ISO 10121-3, ASHRAE Handbook |
| Semiconductor / electronics manufacturing | Airborne molecular contamination (AMC): acids, bases, condensables, dopants | Multi-media systems (carbon + chemisorptive + ion exchange) | ISO 10121-1/-2, SEMI F21 |
| Data centres | SO2, H2S, Cl2, NOx – corrosive to circuit boards and contacts | Impregnated carbon, chemisorptive media | ISO 10121-3, ASHRAE TC 9.9 |
| Industrial workplaces | Welding fumes (organic fraction), solvents, process odours | Activated carbon deep beds, cartridge systems | ISO 10121-1/-2 |
| Airports, tunnels, car parks | NO2, CO, VOCs, kerosene vapours | Activated carbon, catalytic media | ISO 10121-3 |
| Food and beverage production | External odours, cross-contamination of flavours, ethylene | Activated carbon, KMnO4-impregnated media | ISO 10121-1/-2 |
| Nuclear facilities | Radioactive iodine (I-131), noble gases | KI-impregnated activated carbon (TEDA/KI), deep beds | ISO 10121, KTA 3601, IAEA |
HS-Luftfilterbau GmbH supplies a comprehensive range of molecular gas-phase air cleaning products – from activated carbon granules and refillable cartridges to complete V-bank filter systems and chemisorptive media – for all of the above applications.
6. ISO 10121 and Related Standards
ISO 10121 is part of a broader network of standards and guidelines governing air quality and filtration:
| Standard / Guideline | Topic / Relation to ISO 10121 |
|---|---|
| ISO 16890 | Particulate air filters for general ventilation – the particulate counterpart to ISO 10121's gas-phase filtration |
| ISO 29463 / EN 1822 | HEPA and ULPA filters – high-efficiency particulate filtration; ISO 10121 addresses the gas-phase gap that HEPA filters cannot fill |
| Eurovent 4/26 | Eurovent guideline for selection and application of GPACDs based on ISO 10121-3 classification |
| ASHRAE 145.2 | North American standard for laboratory testing of gas-phase air cleaning devices; complementary to ISO 10121 |
| EN 16798-3 | Energy performance of buildings – ventilation requirements including indoor air quality targets for gaseous pollutants |
| ISO 16000 series | Indoor air quality measurement methods – defines the pollutant concentrations that molecular filters are designed to reduce |
| SEMI F21 | Semiconductor industry standard for airborne molecular contamination (AMC) classification and monitoring |
| WHO Air Quality Guidelines | Health-based guideline values for NO2, O3, SO2 and PM – the health driver behind molecular filtration |
| KTA 3601 | German nuclear safety standard – includes requirements for activated carbon iodine filters in nuclear ventilation systems |
7. The Eurovent 4/26 Guideline – Practical Application of ISO 10121-3
Shortly after the publication of ISO 10121-3, the European industry association Eurovent published guideline 4/26, which translates the ISO classification system into practical selection guidance for building designers, HVAC engineers and facility managers. Eurovent 4/26 recommends minimum molecular filter classes based on the ambient pollution level at the building's intake location and the desired indoor air quality target.
This combination of ISO 10121-3 (standardised test and classification) and Eurovent 4/26 (application guidance) provides – for the first time – a complete, manufacturer-independent framework for specifying molecular filtration in ventilation systems. It parallels the established relationship between ISO 16890 (particulate filter classification) and Eurovent 4/23 (particulate filter selection guidance).
8. Activated Carbon in Molecular Filtration: Raw Materials, Quality and Performance
The quality and performance of an activated carbon molecular filter depend critically on the raw material, the activation process and the resulting pore structure. Not all activated carbons are equal – and understanding the differences is essential for selecting the right media for a given application.
| Raw Material | Characteristics | Typical Applications |
|---|---|---|
| Coconut shell | High micropore volume, very hard, low dust, excellent for low-concentration gas adsorption | IAQ, cleanroom AMC, nuclear iodine filtration, drinking water |
| Bituminous coal | Broad pore distribution (micro + meso), good mechanical strength, cost-effective for large volumes | Industrial gas treatment, deep-bed adsorbers, odour control |
| Lignite / brown coal | High mesopore volume, good for larger molecules, lower hardness | VOC abatement, solvent recovery, industrial exhaust |
| Wood | High macropore volume, suitable for liquid-phase and large-molecule applications | Water treatment, decolourisation, food processing |
Key quality parameters for activated carbon in air filtration include: BET surface area (m²/g), iodine number (mg/g), butane activity (%), hardness number (%), ash content (%) and moisture content (%). HS-Luftfilterbau GmbH supplies high-quality activated carbon media from carefully selected raw materials – coconut shell, coal-based and speciality carbons – for refillable cartridges and custom filter systems.


